
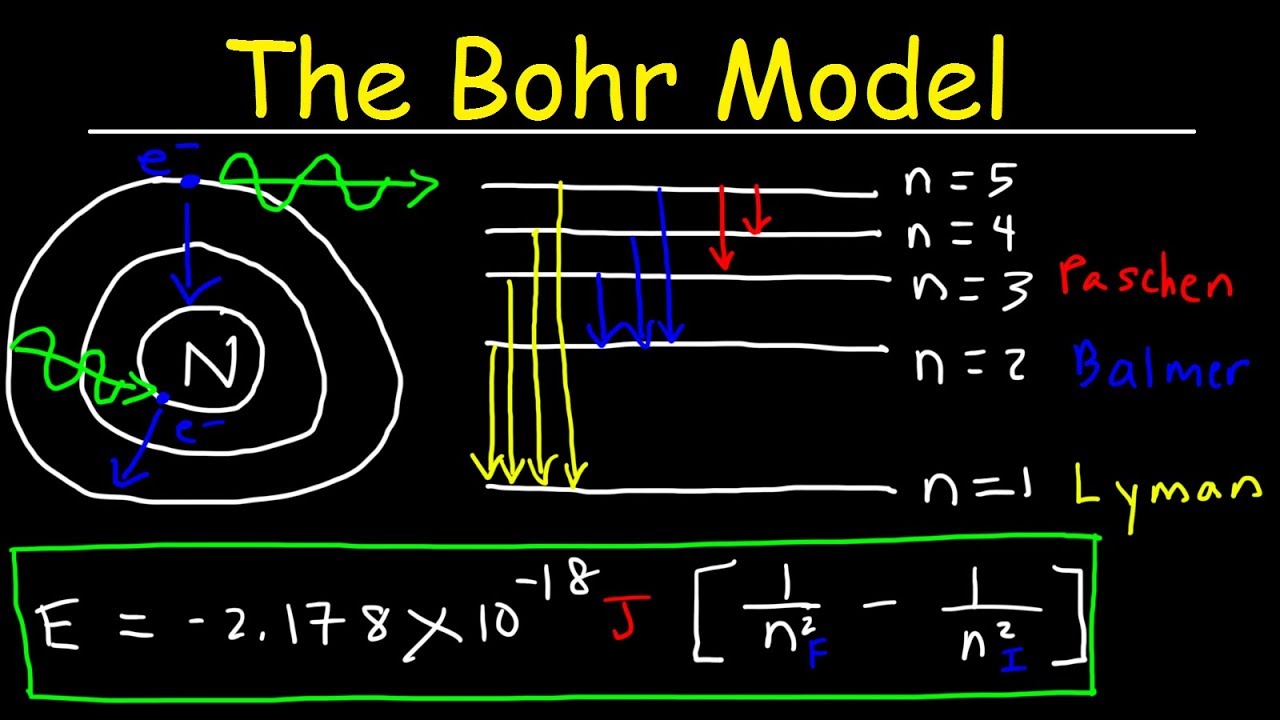
Bohr Model Of The Hydrogen Atom Electron Transitions Atomic Energy Levels Lyman Balmer Series Youtube

The Total Energy Of An Electron In The First Excited State Of Hydrogen Atom Is 3 4 Ev A What Youtube

Energy Of Electron In First Excited State In Hydrogen Atom Is 3 4ev Find Ke And Pe Of Electron Youtube

What Is This Something In Outer Space A Galaxy Perhaps Nope This Is An Atom The Atom Can Be Referred T Quantum Physics Atomic Theory Physics Mechanics

How To Write Electron Configurations For Atoms Of Any Element Electron Configuration Teaching Chemistry Physics And Mathematics












No comments:
Post a Comment