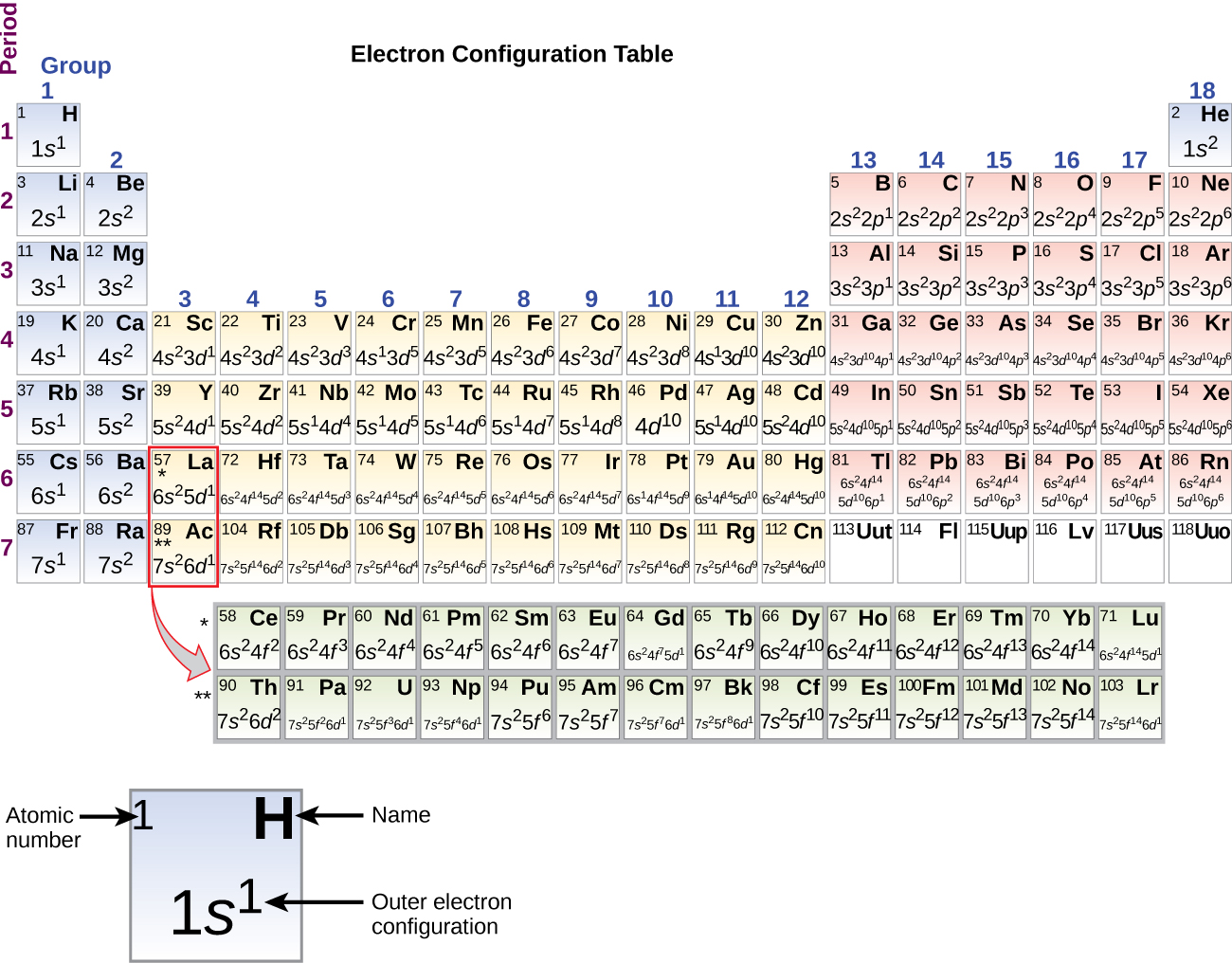
The periodic table is a tabular display of the chemical elements organized on the basis of their atomic numbers electron configurations and chemical properties. Electronic configurations using spectroscopic notation and orbital box notation can be written for elements of atomic numbers 1 to 36.
Electron Configurations How To Write Out The S P D F Electronic Arrangements Of Atoms Ions Periodic Table Oxidation States Using Orbital Notation Gce A Level Revision Notes
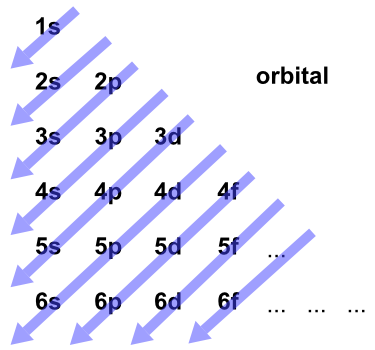
In atomic theory and quantum mechanics an atomic orbital is a mathematical function describing the location and wave-like behavior of an electron in an atom.

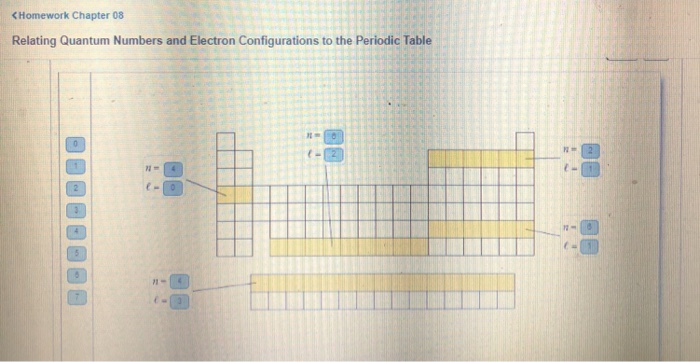
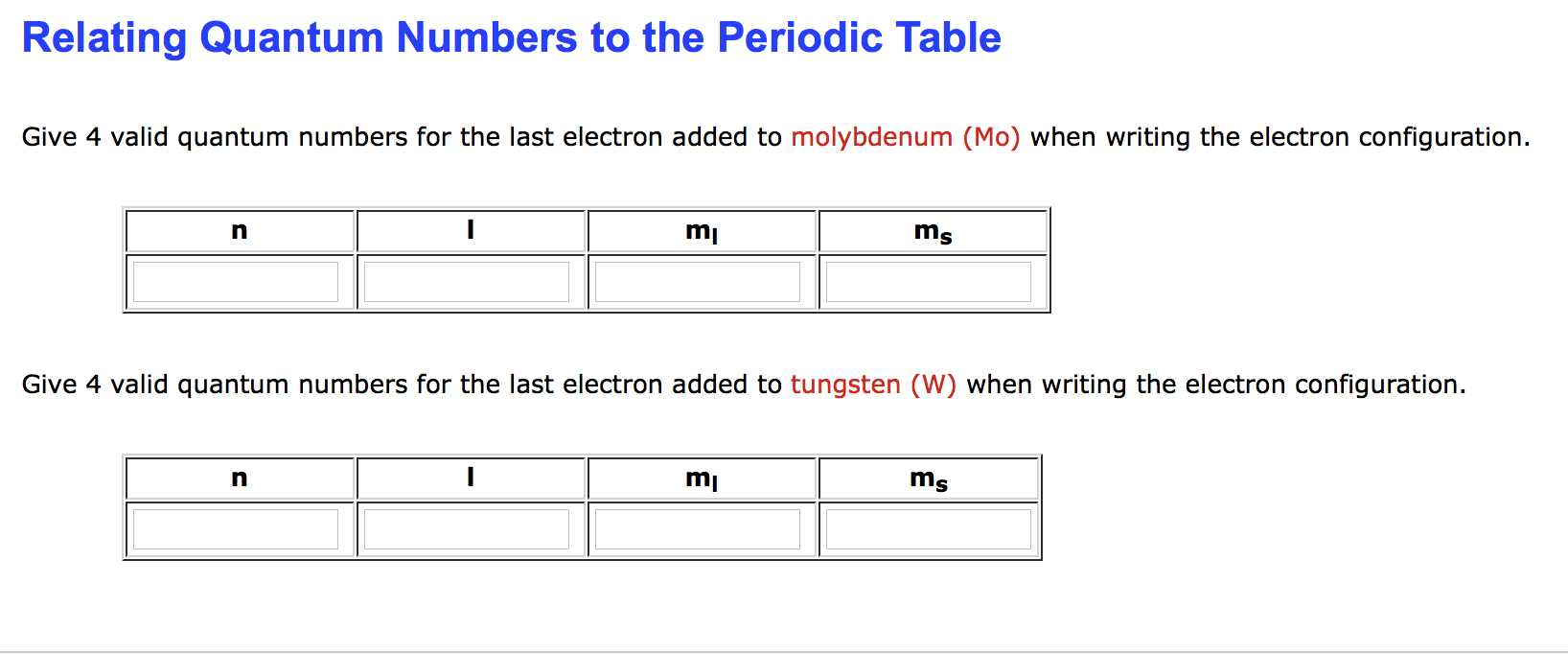
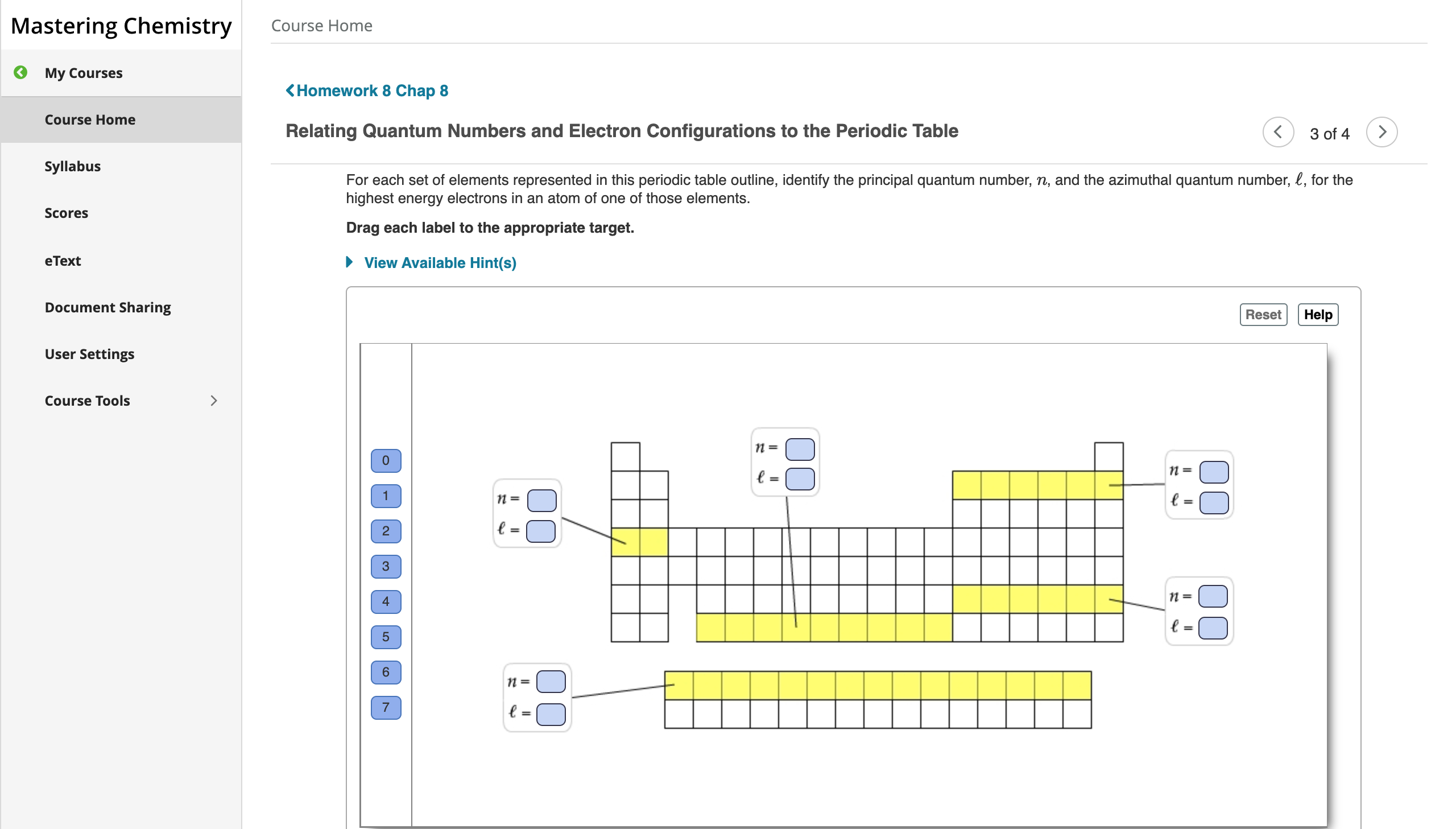
Relating quantum numbers and electron configurations to the periodic table.
A modern periodic table arranges the elements in increasing order of their atomic numbers and groups atoms with similar properties in the same vertical column Figure 2.
This increases the atomic radius as the electrons in the outermost shell are further away from the nucleus.
Cheap paper writing service provides high-quality essays for affordable prices.
The azimuthal quantum number is the second of a set of quantum numbers that describe the unique quantum state of an electron the others being the principal quantum number the magnetic quantum number and the spin quantum number.
It might seem impossible to you that all custom-written essays research papers speeches book reviews and other custom task completed by our writers are both of high quality and cheap.
As we progress down a group in the periodic table the number of electrons increases and so does the number of shells that those electrons are organized into.
The periodic table is a tabular display of the chemical elements organized on the basis of their atomic numbers electron configurations and chemical properties.
All specimens of a pure substance have exactly the same makeup and properties.
Modification of work by vxlaFlickr.
Atomic Radius Definition and Trend.
We write high quality term papers sample essays research papers dissertations thesis papers assignments book reviews speeches book reports custom web content and business papers.
The properties of the elements are periodic functions of their atomic numbers.
Funny pics relating to education.
Modification of work by the Italian voiceFlickr.
Elements are presented in increasing atomic number.
The main body of the table is a 18 7 grid.
The azimuthal quantum number is a quantum number for an atomic orbital that determines its orbital angular momentum and describes the shape of the orbital.
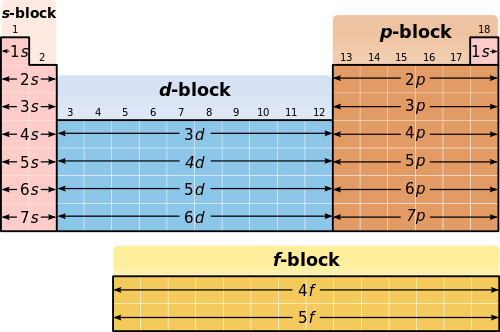
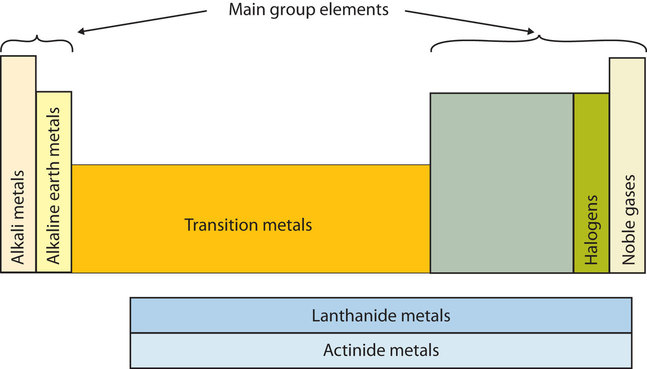
Major Divisions of the Periodic Table.
Isotope Abundance and Atomic Weight The Periodic Table Molecular Formulas and Models Ions and Ion Charges Ionic Compounds Naming Ionic Compounds.
Quantum Numbers and Electron Orbitals.
Periodic Table Projects Experiments Scientific Method Biochemistry Physical Chemistry.
Quantum Numbers and Electron Configurations Electron Configurations of Atoms and Ions and Magnetic.
Any sample of sucrose table sugar consists of 421 carbon 65 hydrogen and 514 oxygen by mass.
Learn Something New Every Day.
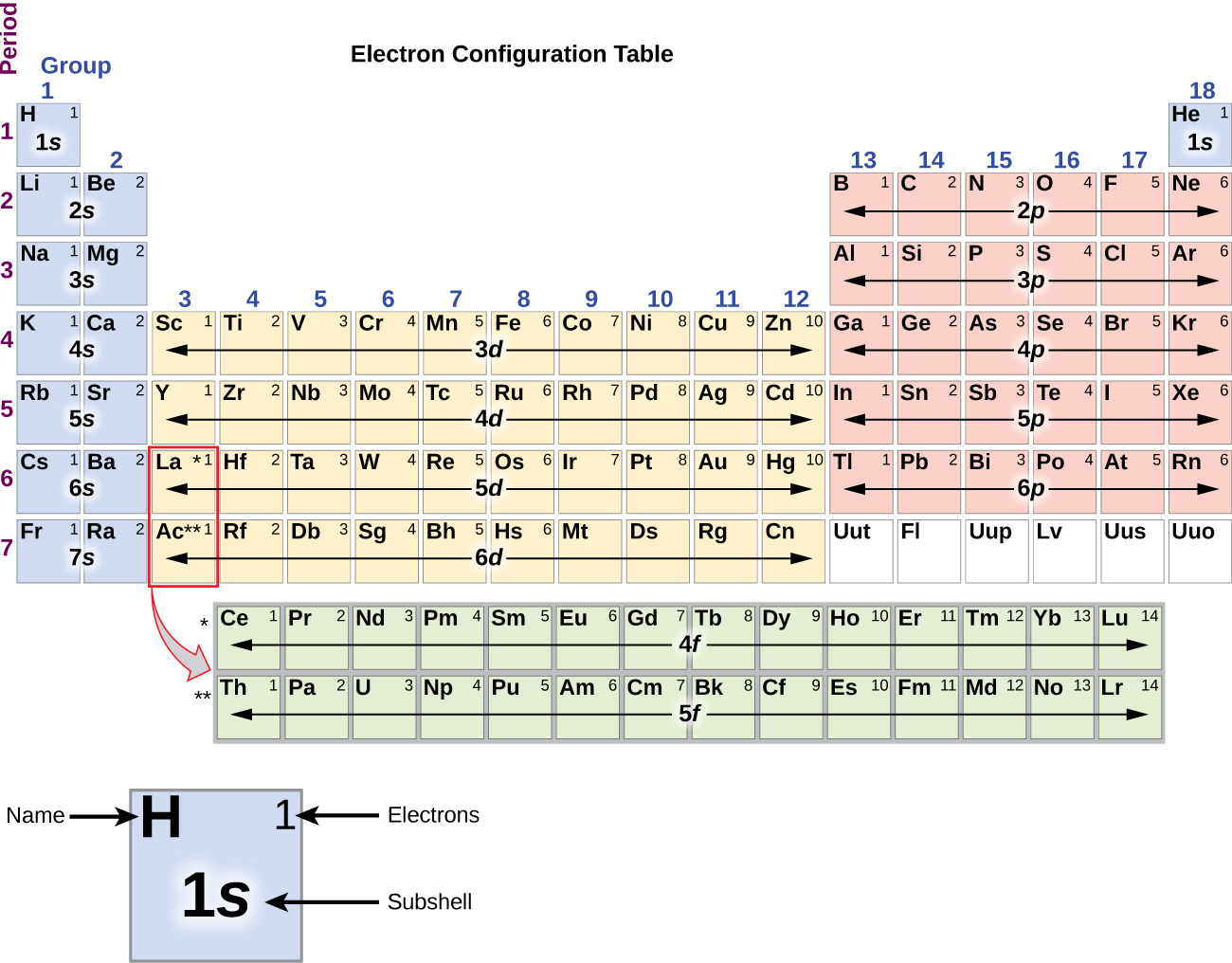
Relating Shells Subshells and Orbitals.
Elements are presented in increasing atomic number.
The modern statement of this relationship the periodic law is as follows.
Figure 11 Chemical substances and processes are essential for our existence providing sustenance keeping us clean and healthy fabricating electronic devices enabling transportation and much more.
Modification of work.
The main body of the table is a 18 7 grid.
Atomic Radius Trends on the Periodic Table.
This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atoms nucleusThe term atomic orbital may also refer to the physical region or space where the electron can be.
The energy can be released as one quantum of energy as the electron returns to its ground state say from n 5 to n 1 or it can be released as two or more smaller quanta as the electron falls to an intermediate state then to the ground state say from n 5 to n 4 emitting one quantum then to n 1 emitting a second quantum.
The periodic table is subdivided into four blocks s p d and f corresponding to the outer.
Using orbital box notation for the first four shells of a multi-electron atom.
Any sample of sucrose also has the same physical properties such as melting point color and sweetness regardless of the source from which it is isolated.
The latest Lifestyle Daily Life news tips opinion and advice from The Sydney Morning Herald covering life and relationships beauty fashion health wellbeing.
School Humor Play Classic Asteroids.
Https Encrypted Tbn0 Gstatic Com Images Q Tbn And9gcseie3ypz 5dcsjpxr2y0l Qzja6r6ypbvsahw0bslrdcaafqq Usqp Cau

Electron Configurations Using Periodic Table Explained In Easy Tutorial Crash Chemistry Academy Youtube













No comments:
Post a Comment